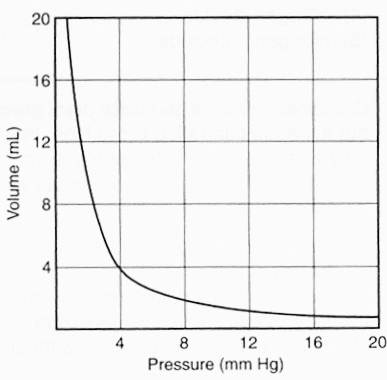
This graph represents the relationship of the pressure and volume of a given mass of a gas at constant temperature. When the pressure equals 8 millimeters of mercury (mmHg), what is the volume, in milliliters (mL)?
Explanation
Locate the given pressure, 8 mm, along the horizontal axis. Move up the 8 mm line until the graph curve is reached. On the vertical axis at the left, you will find that the volume at this point is approximately 2 mL.
Visit our website for other GED topics now!
Related questions