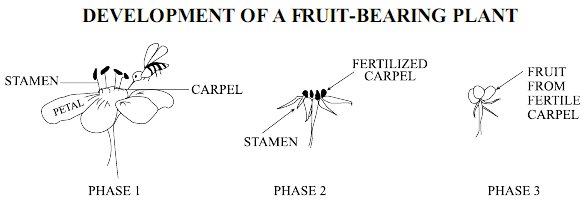
Isomers are molecules that have the same number of the same elements, but in a different arrangement.Which one of the following is an isomer of 2-propanol (shown below), which contains Carbon (C), Hydrogen (H) and Oxygen (O)?

Explanation
According to the molecular structure provided in the question, 2-propanol has three carbon atoms, eight hydrogen atoms, and one oxygen atom. Only the structure in choice b has the same number of each of those atoms, but in a different arrangement (oxygen is attached to a different carbon),making the structure in choice b, an isomer of 2-propanol.
Visit our website for other GED topics now!
Related questions